|
The second product, Na 2CrO 4, also adheres to Rule #1, which states that salts of alkali metals tend to be soluble.īecause both products are soluble, no precipitate form as a result of this reaction. Therefore, this compound will not contribute to any precipitation being formed. Although Rule #5 says that hydroxides tend to be insoluble, Rule #1 states that salts of alkali metal cations tend to be soluble, and Rule #1 precedes Rule #5. The first product, KOH, is an example of two rules contradicting each other. 
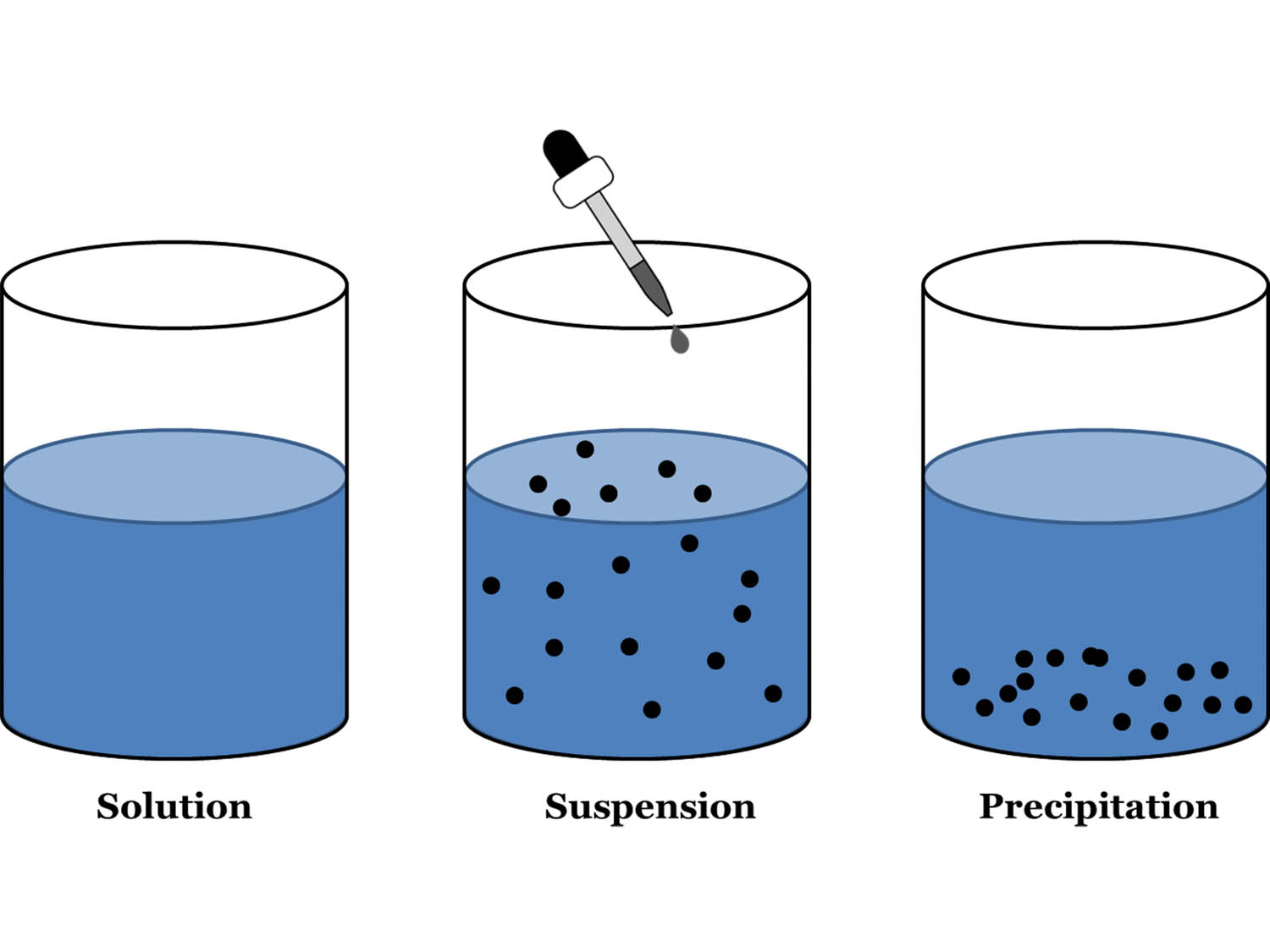
Predict if a precipitate will form as a result of this reaction:Ĭonsider again the products of the reaction: if either is insoluble, a precipitate will form. Therefore, because of this compound, a precipitate will form in the course of this reaction.ĥ. According to Rule #5, that sulfides tend to be insoluble. A precipitate of this compound will not form. The products of the reaction must be examined if either of the substances formed in the reaction is insoluble, a precipitate will form.Ĭonsidering NaNO 3, Rule #3 states that nitrates tend to be soluble. Predict whether a precipitate will form as a result of this reaction: Because Rule #3 precedes Rule #4, the compound is insoluble and will form a precipitate.Ĥ. The reaction mechanism involves the reaction between dissolved metals in aqueous waste and precipitants to produce fine insoluble. Rule #4 states that bromides are usually soluble, but Rule #3 states that salts of silver are insoluble. Chemical precipitation is the most commonly used method due to the ease of handling and low-cost for the treatment of toxic heavy metals dissolved in industrial effluent before it is discharged ( Li et al., 2003 ). Letters a and c are both likely to form precipitates.Ĭoncerning a) CaSO 4, although sulfates tend to be soluble, Rule #5 indicates that calcium sulfate is an important exception to this rule.įor b), Rule #1 indicates that table salt (NaCl) is soluble because it is a salt of an alkali metal.Ĭ) is an example of two rules contradicting each other. Which of these substances is likely to form a precipitate? Therefore, it will not form a precipitate.ģ. This is perchlorate, which according to Rule #2 is likely to be soluble. Therefore, FeCO 3 is likely to form a precipitate.Ģ. Fluorides such as BaF 2, MgF 2, and PbF 2 are frequently insoluble.Īccording to Rule #5, carbonates tend to be insoluble.Phosphates such as Ca 3(PO 4) 2 and Ag 3PO 4 are frequently insoluble. 
Group II carbonates (CaCO 3, SrCO 3, and BaCO 3) are insoluble, as are FeCO 3 and PbCO 3. Strong electrolyte (definition) An electrolyte that exists in solution almost entirely as ions. Nonelectrolyte (sample) Sucrose (C12H22O11) Molecules (charge) Neutral. States of matter must be included in the chemical equation for a precipitation reaction. Arsenic, antimony, bismuth, and lead sulfides are also insoluble. Hydrogen Chloride gass (HCl (g)) Nonelectrolyte (definition) A substance that dissolves in water to give a nonconducting or very porrly conducting solution. definition of an acid is that it is a hydrogen ion donor.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
